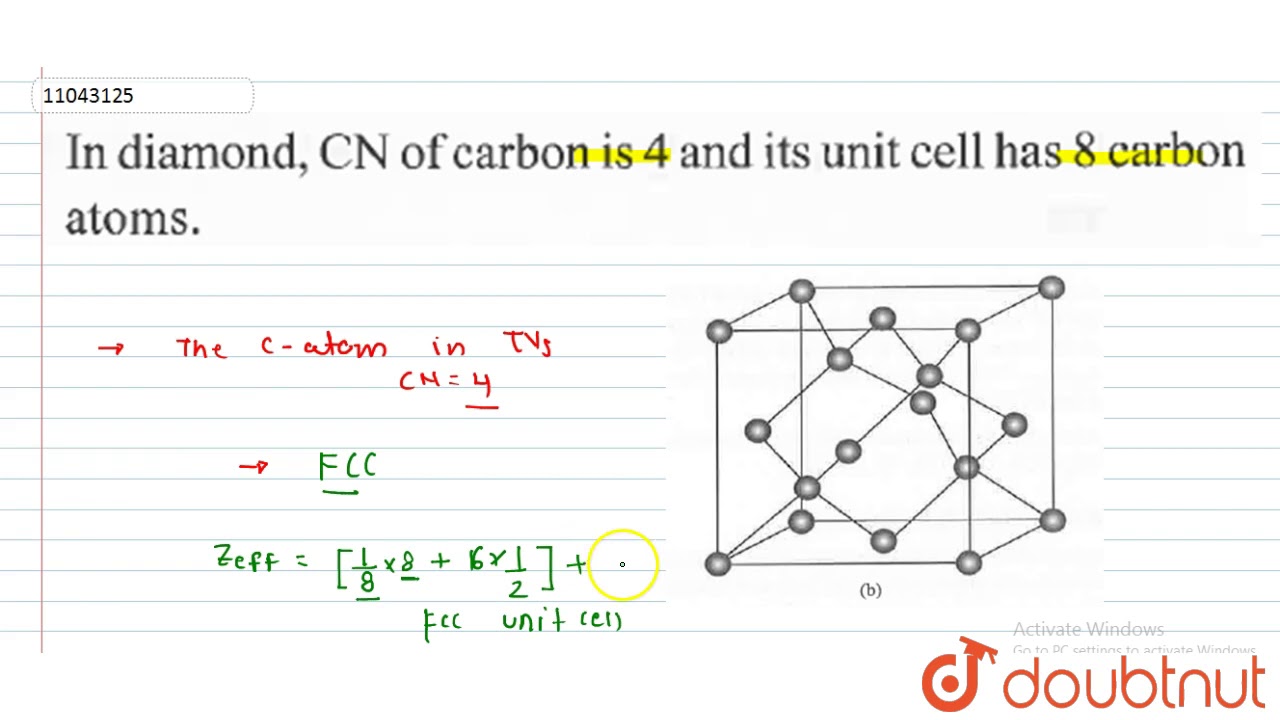
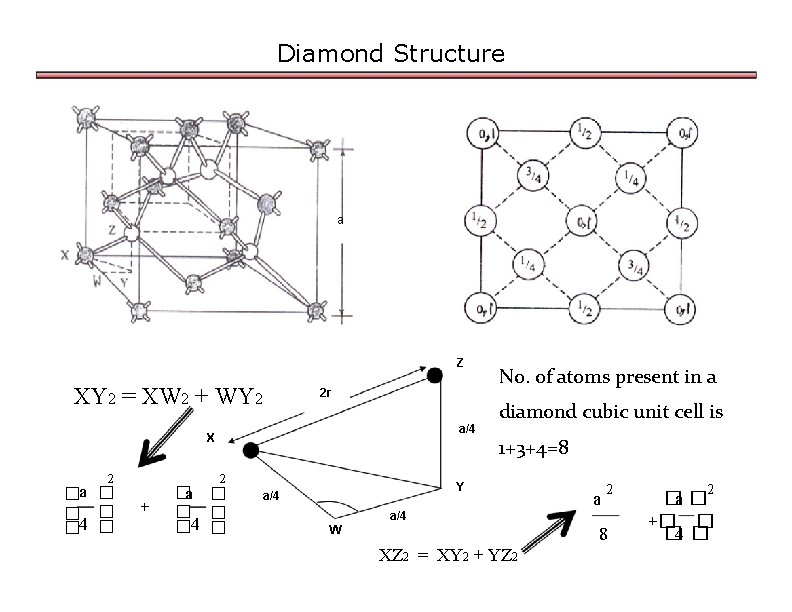
In a crystal of diamond:(i) How many carbon atoms are present per unit cell?(ii) What type of lattice does diamond crystallize in?(iii) How many carbon atoms surround each carbon atom?(iv) How are

If one mole of carbon atoms weighs 12 grams, what is the mass (in grams) of 1 atom of carbon?... - YouTube

How many atoms of carbon are present in a diamond weighing 0.5 carat? (one carat weighs 200 mg and - Brainly.in

Diamond has a zinc blende type structure in which carbon atoms form CCP lattice and carbon atoms occupy tetrahedral voids. If the edge length of the cube is 3.6 Å then the

OneClass: what is the average spacing between carbon atoms in diamond, density of which is 3.51g/cm^3